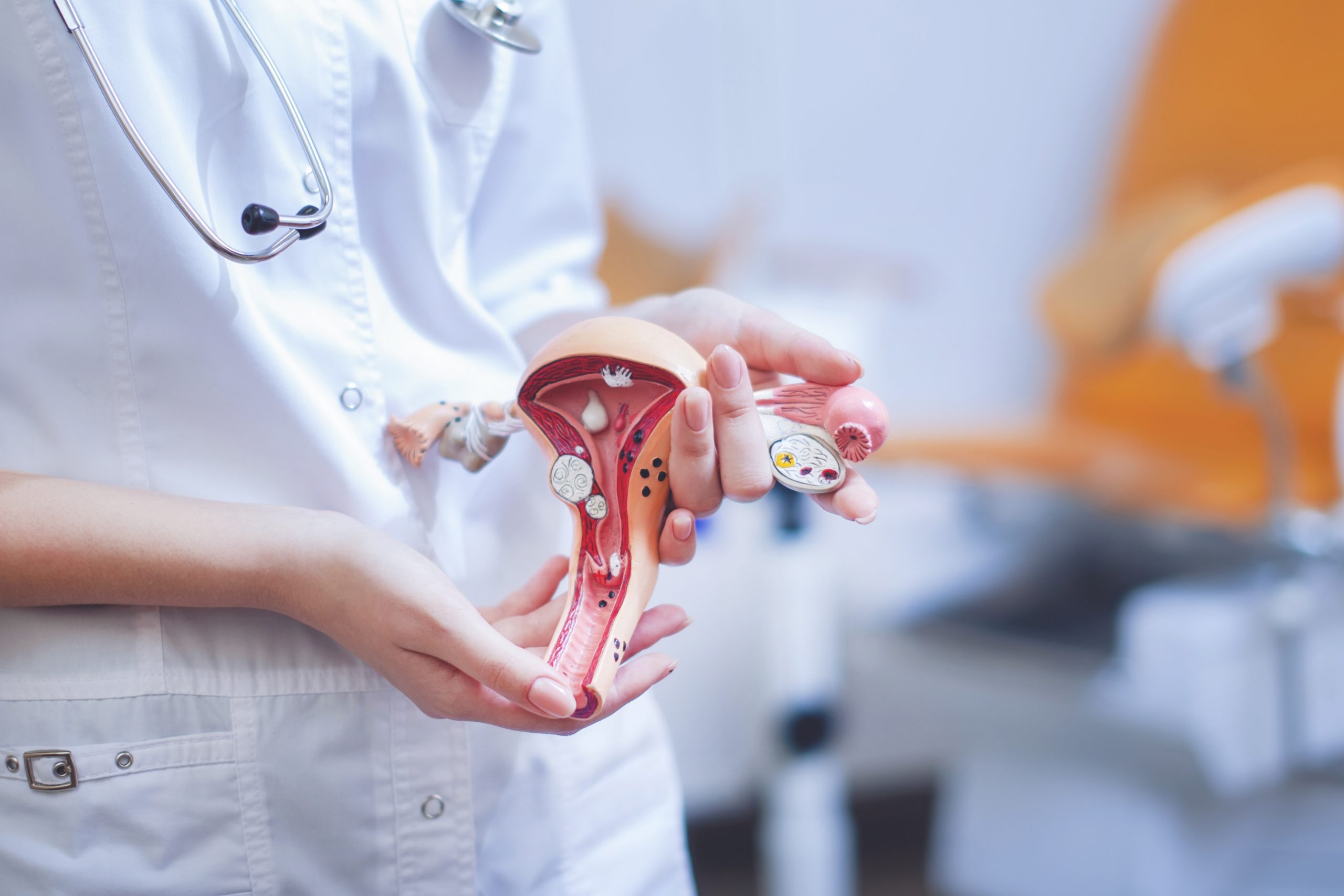
I nostri percorsi e trattamenti

Probabilità di successo
Il successo di un trattamento di PMA dipende da molteplici elementi: competenza, tecnologia, fattore umano.
Consulta qui i tassi di successo dei trattamenti dei pazienti che si affidano a noi.
Perché scegliere Genera
La nostra Scienza e la migliore tecnologia per ottenere una nascita.
Protocolli di cura personalizzati per ogni paziente
Leader in ricerca scientifica
Laboratori di embriologia avanzati
Alti tassi di successo rispetto alla media nazionale
Zero liste di attesa, hai subito il tuo appuntamento
Banca di ovociti interna al gruppo
8
centri specializzati
+500
bimbi nati ogni anno
30
anni di esperienza
+3000
trattamenti ogni anno

Le nostre cliniche
Genera è una rete di cliniche specializzate in procreazione assistita. Siamo presenti in tutta Italia con centri di I, II e III livello. Tutti i nostri centri sono dotati di tecnologia all’avanguardia e di personale altamente qualificato, la migliore combinazione per supportare i pazienti in ogni fase del percorso di cura.
Equipe Genera
Conosci la PMA
L’informazione multimediale dedicata ai pazienti dei centri Genera e a chiunque sia alla ricerca di approfondimenti, analisi e curiosità sulla Procreazione Medicalmente Assistita.